Studies on the Synthesis of Perfluoroaryl Sulfides and their Application in Desulfurative Nickel-Catalyzed Reductive Cross- Coup

Molecules | Free Full-Text | How Do Aromatic Nitro Compounds React with Nucleophiles? Theoretical Description Using Aromaticity, Nucleophilicity and Electrophilicity Indices
Synthesis of perfluoroaryl sulfides at electron-poor arenes via an SNAr step with an unexpected mechanism

First-principles study of the reaction mechanism governing the SNAr of the dimethylamine on 2-methoxy-5-nitrothiophenes | SpringerLink

Molecules | Free Full-Text | How Do Aromatic Nitro Compounds React with Nucleophiles? Theoretical Description Using Aromaticity, Nucleophilicity and Electrophilicity Indices
How Do Aromatic Nitro Compounds React with Nucleophiles? Theoretical Description Using Aromaticity, Nucleophilicity and Electrop
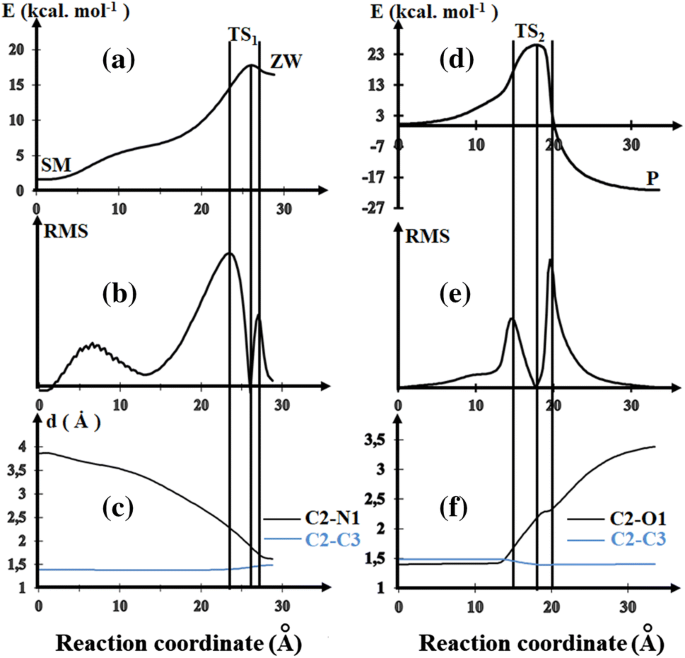
First-principles study of the reaction mechanism governing the SNAr of the dimethylamine on 2-methoxy-5-nitrothiophenes | SpringerLink

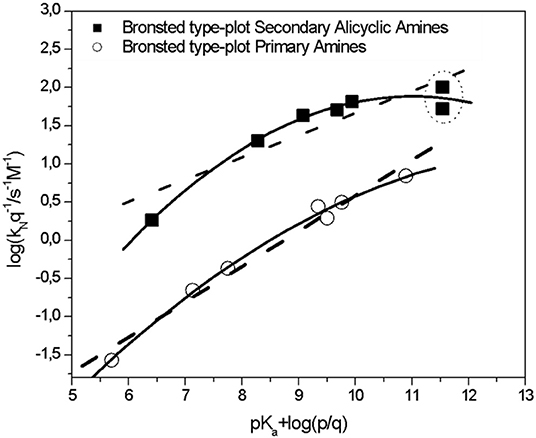
Reaction mechanism of the SNAr reaction between 1-X-2,4-dinitrobenzene... | Download Scientific Diagram
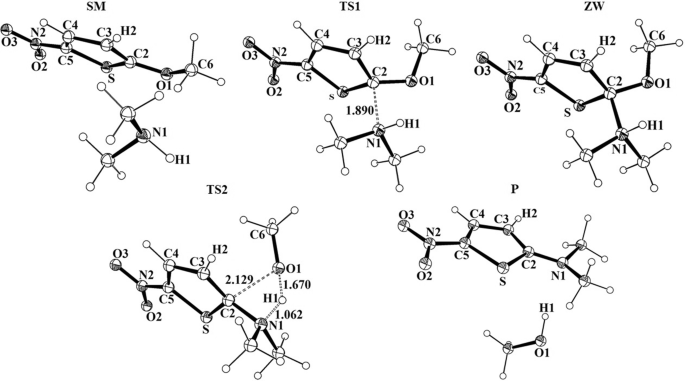
First-principles study of the reaction mechanism governing the SNAr of the dimethylamine on 2-methoxy-5-nitrothiophenes | SpringerLink
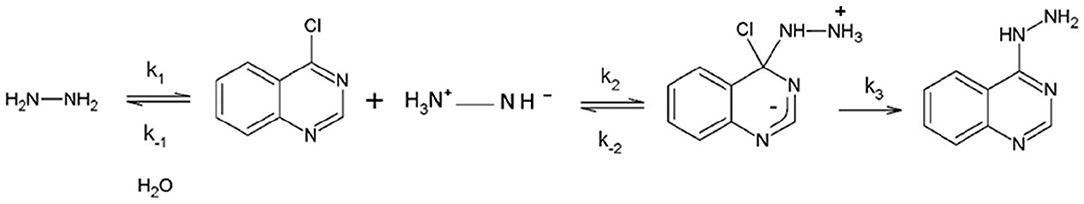
Frontiers | Activation of Electrophile/Nucleophile Pair by a Nucleophilic and Electrophilic Solvation in a SNAr Reaction

Meisenheimer complexes as hidden intermediates in the aza-S N Ar mechanism - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D0OB00600A

Secreted reporter assay enables quantitative and longitudinal monitoring of neuronal activity | bioRxiv

Unusual Spectroscopic and Electric Field Sensitivity of Chromophores with Short Hydrogen Bonds: GFP and PYP as Model Systems | The Journal of Physical Chemistry B
Studies on the Synthesis of Perfluoroaryl Sulfides and their Application in Desulfurative Nickel-Catalyzed Reductive Cross- Coup