IJMS | Free Full-Text | Molecular Cloning and Exploration of the Biochemical and Functional Analysis of Recombinant Glucose-6-Phosphate Dehydrogenase from Gluconoacetobacter diazotrophicus PAL5

Amazon.com : Miracle Plus Arnica Bruise Relief Gel - Natural Skin Care for Bruising, Swelling & Discoloration, 4 Fl Oz : Health & Household
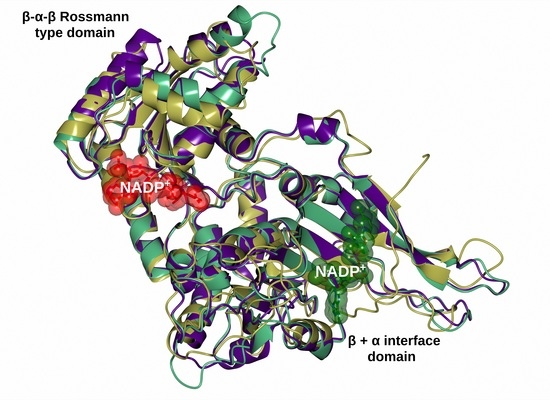
Biomolecules | Free Full-Text | S-adenosyl-l-homocysteine Hydrolase: A Structural Perspective on the Enzyme with Two Rossmann-Fold Domains
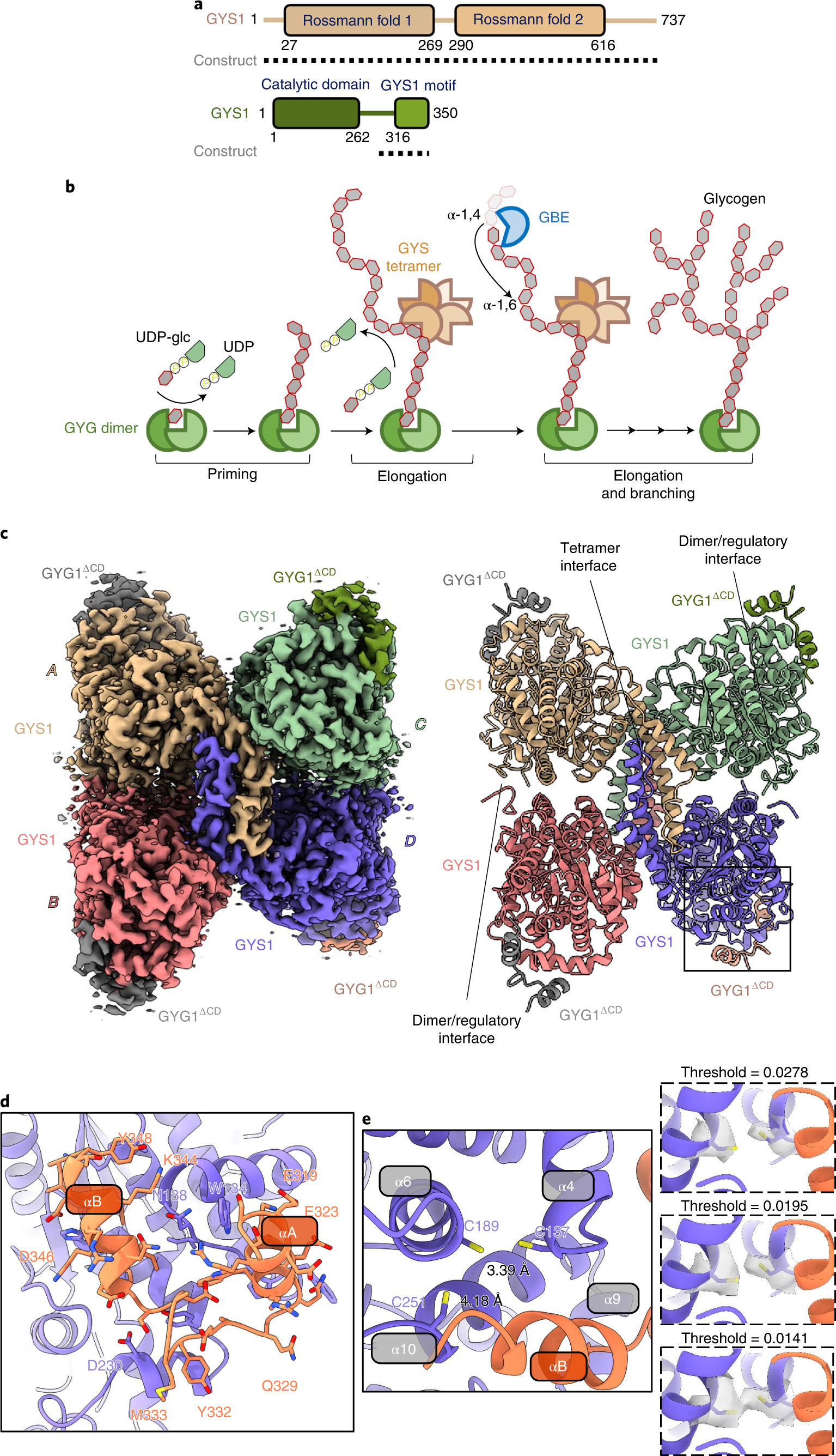
Molecular basis for the regulation of human glycogen synthase by phosphorylation and glucose-6-phosphate | Nature Structural & Molecular Biology
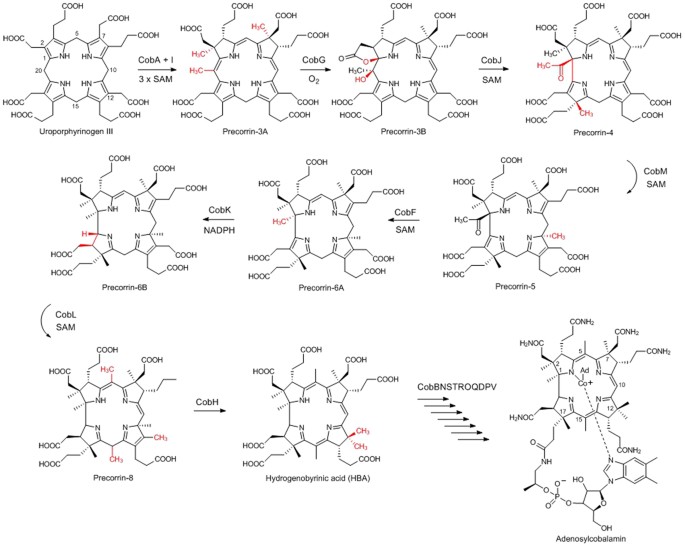
Crystal structure of CobK reveals strand-swapping between Rossmann-fold domains and molecular basis of the reduced precorrin product trap | Scientific Reports

Rossmann-Fold Methyltransferases: Taking a “β-Turn” around Their Cofactor, S-Adenosylmethionine | Biochemistry
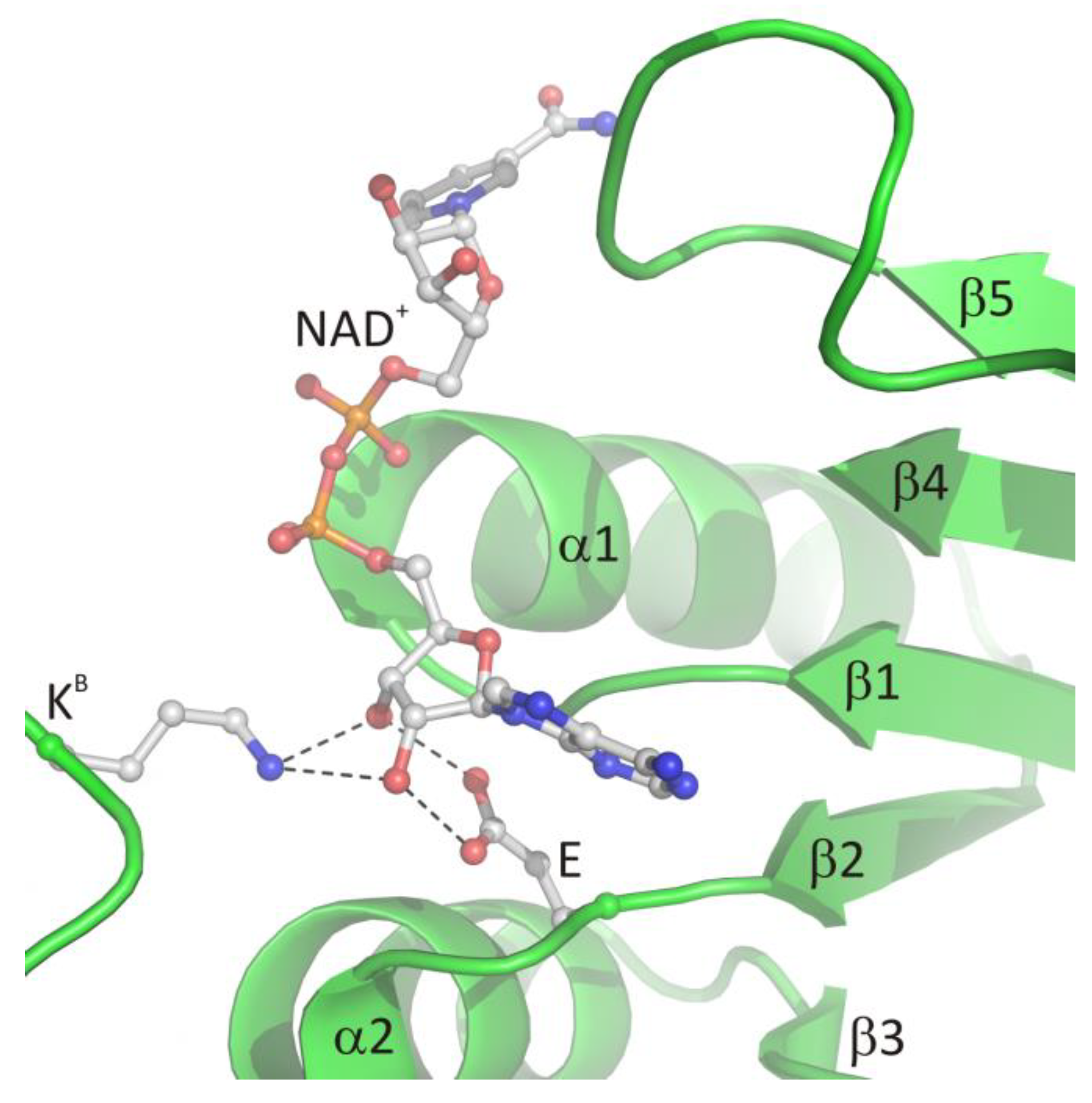
Biomolecules | Free Full-Text | S-adenosyl-l-homocysteine Hydrolase: A Structural Perspective on the Enzyme with Two Rossmann-Fold Domains

Rossmann-Fold Methyltransferases: Taking a “β-Turn” around Their Cofactor, S-Adenosylmethionine | Biochemistry